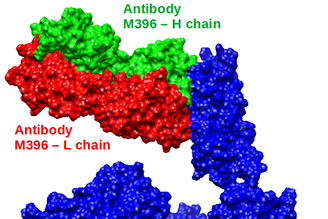
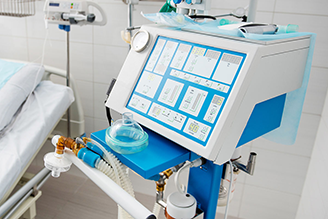
Lawrence Livermore National Laboratory (LLNL) is fully committed to helping protect the U.S. from COVID-19 and to speed the recovery of those affected. As a world-class research institute, we have considerable infrastructure, unique research capabilities and a dedicated team of scientists and engineers supporting the fight against the COVID-19 pandemic.
Since the emergence of the pandemic, we have been supporting the research and medical communities with tools and information to better understand COVID-19. For example, we released predicted structures of a key COVID-19 viral protein to help the research community accelerate development of potential medical treatments. A summary of our initial research efforts is also available, as is information about our participation in the COVID-19 High Performance Computing Consortium. LLNL is also a part of the Department of Energy’s collective response to the pandemic.
The Laboratory is a highly collaborative organization, and we welcome partnerships with academia and industry. Our COVID-19 research and response activities are focused on four broad areas: High Performance Computing, Detection, Medical Countermeasures and Medical Equipment. The Laboratory is also leveraging our unique capabilities and infrastructure to support the fight against COVID-19, as well as creating and maintaining a COVID-19 data portal to expedite access to our modeling results. Our Innovation and Partnerships Office has identified technologies and capabilities that are currently available under free, temporary non-exclusive licenses. For technical inquiries and collaborative opportunities, please contact the Laboratory’s Biosecurity Center at biosecurity [at] llnl.gov (biosecurity[at]llnl[dot]gov). More information about our current activities can be found on the following pages.
This video summarizes some of the work that Lawrence Livermore researchers and staff are doing to respond to the COVID-19 epidemic.
COVID-19 Data Portals
Provisioning access to scientific data via interactive data querying and visualization systems.
- Accelerated publication - High-Throughput Virtual Screening of Small Molecule Inhibitors for SARS-CoV-2 Protein Targets with Deep Fusion Models
- Publication - Quantitative Fit Evaluation of N95 Filtering Facepiece Respirators and Coronavirus Inactivation Following Heat Treatment
- Accelerated publication - Rapid in silico design of antibodies targeting SARS-CoV-2 using machine learning and supercomputing
- June 30, 2022 - UC Merced students work with LLNL mentors on potential new drugs to combat COVID-19
- March 10, 2022 - Machine learning model finds COVID-19 risks for cancer patients
- February 15, 2022 - LLNL team models COVID-19 disease progression and identifies risk factors
- December 1, 2021 - Transferring Laboratory technology to fight the COVID-19 pandemic
- October 21, 2021 - LLNL joins Human Vaccines Project to accelerate vaccine development and understanding of immune response
- October 11, 2021 - Tackling the COVID-19 pandemic
- June 2, 2021 - Decontaminating N95 masks for reuse
- May 3, 2021 - Enabling rapid COVID-19 small molecule drug design through scalable deep learning of generative models
- April 1, 2021 - COVID-19 HPC Consortium reflects on past year
- February 19, 2021 - Physics of particle dispersion may lend insight into reducing the airborne spread of COVID-19 virus
- January 19, 2021 - LLNL and United Kingdom company to collaborate on development of universal coronavirus vaccine
- November 18, 2020 - Model for COVID-19 drug discovery a Gordon Bell finalist
- November 12, 2020 - LLNL welcomes 'Ruby' supercomputer for national nuclear security mission and COVID-19 research
- November 4, 2020 - Mammoth computing cluster to aid COVID research
- October 19, 2020 - AI gets a boost via LLNL, SambaNova collaboration
- October 7, 2020 - Corona supercomputer gets funding for COVID-19 work
- June 25, 2020 - Lab technologies help fight COVID-19
- June 9, 2020 - Lab, BMI produce FDA-approved emergency ventilator
- June 8, 2020 - Multi-lab research to improve COVID-19 diagnostics
- May 29, 2020 - Lab team studies calibrated AI and deep learning models to more reliably diagnose and treat disease
- May 21, 2020 - Lab provides assistance in national swab shortage
- May 19, 2020 - COVID-19 research goes public through new portal
- May 14, 2020 - Deactivating coronavirus on N95 respirators for reuse
- May 1, 2020 - LLNL’s new machine learning platform generates novel COVID-19 antibody sequences for experimental testing
- April 29, 2020 - LLNL develops ‘stopgap’ ventilator for COVID-19 use
- April 21, 2020 - Upgrades for LLNL supercomputer from AMD, Penguin Computing aid COVID-19 research
- March 26, 2020 - New partnership to unleash U.S. supercomputing resources in the fight against COVID-19
- March 26, 2020 - Lab antibody, anti-viral research aids COVID-19 response
- February 3, 2020 - Lawrence Livermore researchers release 3D protein structure predictions for the novel coronavirus
- April 21, 2021 - Corona vs coronavirus: AMD and Penguin Computing to upgrade aptly named LLNL supercomputer
- April 1, 2021 - Beating back the coronavirus requires a bigger arsenal
- March 26, 2021 - The COVID-19 HPC Consortium looks ahead to a ‘National Strategic Computing Reserve’
- March 22, 2021 - What is a universal coronavirus vaccine and is it achievable?
- February 21, 2021 - 'This has to be the moment' to invest in coronavirus vaccines and treatments against future pandemics, experts warn
- February 11, 2021 - Worries About Viral Resistance to Covid-19 Vaccines Are Overdone
- January 13, 2021 - Why There's Still Need for Rapid Accessible Pathogen Testing, During and After the Crisis
- November 4, 2020 - AMD-Supermicro-Cornelis (Omni-Path) ‘Mammoth’ cluster at LLNL targets COVID-19
- May 5, 2020 - Advanced manufacturing innovation helps industry in COVID-19 fight
- May 5, 2020 - 3-D printed COVID-19 test swabs pass their own tests
- May 4, 2020 - Swab Shortage: FATHOM & Abiogenix Bridge the Gap with 3D Printed NP Swabs
- April 14, 2020 - LLNL’s Jim Brase explains how DOE labs are fighting (and coping with) COVID-19
- February 5, 2020 - Coronavirus: Lawrence Livermore Lab researchers examine virus in hopes of blocking, treating it
For more information about the U.S. government's response to COVID-19, see coronavirus.gov and usa.gov/coronavirus. For the latest public health and safety information from the Centers for Disease Control and Prevention, see cdc.gov/coronavirus.