Lab technologies help fight COVID-19
 (Download Image)
(Download Image)
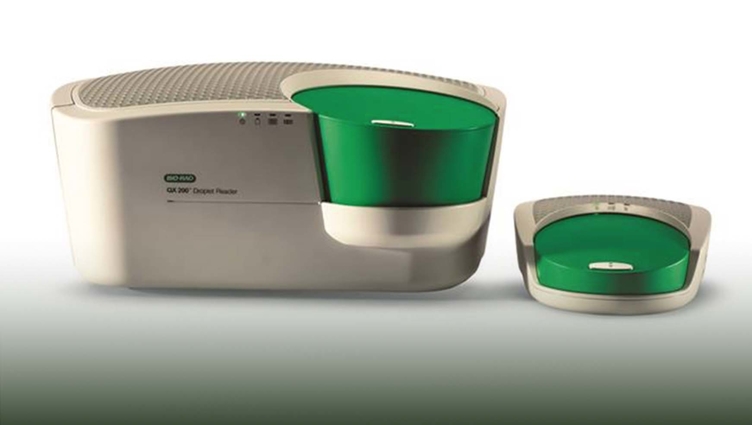
Used by researchers around the world, Bio-Rad's QX200 Droplet Digital PCR (ddPCR) System quantifies target DNA or RNA molecules. The company’s SARS-CoV-2 Droplet Digital PCR (ddPCR) test kit is run on the QX200.
Lawrence Livermore National Laboratory’s (LLNL) technology transfer team has opened up multiple fronts to aid the nation’s efforts against the SARS-CoV-2 virus that causes COVID-19.
One Bay Area company has developed a test to detect SARS-CoV-2 based on an LLNL technology while a second Bay Area firm that has licensed an LLNL technology has gained approval for a product to diagnose COVID-19.
In addition, the office of the LLNL Deputy Director for Science and Technology has invested more than $1 million to develop a prototype ventilator and other technologies, and the Lab’s Innovation and Partnerships Office (IPO) is granting royalty-free, non-exclusive licenses for LLNL technologies that could be used against the pandemic.
“Partnerships are integral to much of what we do at LLNL,” said Rich Rankin, the Lab’s IPO director. “This reality is especially driven home when there is a crunch, when there’s a crying need for a science solution to a problem.
“We conduct long-term research for solutions to national problems, year in and year out, but LLNL shines when there’s a critical need for solutions to important challenges. That’s something within the DNA of the Laboratory.”
In early May, Bio-Rad Laboratories Inc., a global leader of life science research and clinical diagnostics products, announced that its SARS-CoV-2 Droplet Digital PCR (ddPCR) test kit had been granted emergency use authorization by the U.S. Food and Drug Administration (FDA).
The SARS-CoV-2 ddPCR test runs on Bio-Rad’s QX200 and QXDx ddPCR systems. The test is based on a technology that was licensed from LLNL more than 10 years ago.
The high sensitivity of the Bio-Rad test makes it well-suited to screening upper respiratory samples in patients with a low viral load, including individuals in the early stages of infection as compared to classical quantitative PCR tests.
The test also can play an important role in surveillance by detecting minimal residual disease in people recovering from COVID-19, informing them if they are negative for the virus. Bio-Rad’s single-well SARS-CoV-2 ddPCR test provides clinicians with a high degree of sensitivity that can significantly improve the accuracy of reported results.
Cepheid Inc., the other LLNL licensee that has received an emergency use authorization from the FDA for SARS-CoV-2 diagnostic tests, licensed a technology called rapid PCR thermocycling, integrating amplification and detection.
Co-founded in 1996 by former LLNL researcher Allen Northrup, Cepheid had its detection technology used by Northrup Grumman after the 2001 anthrax attacks to screen mail for bacteria. It also was deployed for the detection of Ebola.
On a second front, the Laboratory’s IPO has used more than $1 million of licensing and royalty money, produced by Lab licenses, to fund the development of a mechanical ventilator and research on other COVID-19-related technologies.
Funds are received from the licensing of technologies developed by LLNL scientists and engineers to companies that create commercial products and then pay LLNL royalties when products are sold.
“In effect, LLNL researchers create inventions that others commercialize and return a fraction of their revenues to the Laboratory. These funds can be used in crises like the COVID-19 pandemic to conduct targeted research and development. It’s something of a virtuous cycle,” Rankin said.
The LLNL mechanical ventilator’s design is derived from proven concepts and contains parts that are not being used by commercial ventilator manufacturers, to avoid disrupting already thin supply chains.
The ventilator, branded as SuppleVent™, has received an emergency use authorization from the FDA and is the focus of a cooperative research and development agreement (CRADA) with North Carolina-based BioMedInnovations LLC. The CRADA was concluded in less than 10 days, once the deal’s scope was defined.
Another project under way with IPO funds, which are dispersed through the office of the Lab’s Deputy Director for Science and Technology, is a diagnostic tool.
LLNL biomedical researcher Larry Dugan is seeking to develop a simple, one-tube sample-to-result diagnostic to detect SARS-CoV-2 in oral and nasal samples in under 60 minutes. This diagnostic would provide medical professionals and emergency responders with a non-invasive, highly sensitive detection tool.
On a third front, the IPO has identified LLNL technologies and capabilities that could be used by industry to respond to the crisis by making the technologies available under non-exclusive, royalty-free, time-limited licenses.
“The thought behind it is to help companies to evaluate a new technology for addressing an aspect of COVID-19 at a lower risk,” Rankin said. “As the crisis passes, the company can come and look at possible longer-term licenses.”
Rankin emphasized that IPO doesn’t only seek to build external partnerships with commercial ties, but to forge close partnerships with organizations within LLNL.
“During the latest pandemic, we are and have been working very closely with the Livermore Field Office of the Department of Energy, Legal, Engineering, Physical and Life Sciences, Global Security, the Chief Financial Officer, the Office of Classification and Export Control and others across the Lab.
“Each of these organizations are either conducting research or they ensure that our partnership agreements are sound. It is our desire to use technologies developed at LLNL to help protect the health and safety of people in this country and around the world,” Rankin said.
Note: Bio-Rad, Droplet Digital PCR and ddPCR are trademarks of Bio-Rad Laboratories, Inc. in certain jurisdictions.
Contact
 Stephen Wampler
Stephen Wampler
[email protected]
(925) 423-3107
Related Links
LLNL Innovation and Partnerships OfficeBio-Rad Laboratories Inc.
Cepheid Inc.
“Lab, BMI produce FDA-approved emergency ventilator"
Tags
Industry CollaborationsTechnology Transfer
Featured Articles







