LAB REPORT
Science and Technology Making Headlines
Dec. 6, 2019

When asteroids break up, they leave a telltale trail of dusty debris in their wake. Image by NASA/JPL-Caltech
Asteroid assassination
At first astronomers thought they had spotted a comet, but it really was an asteroid in the belt between Mars and Jupiter being struck by another object.
The asteroid belt is not like the cluttered debris field in “The Empire Strikes Back.” It may contain millions of rocky and metal objects, but the distances separating them are vast and collisions are rare.
That is what makes P/2016 G1 such an exciting object. Spotted zipping through the asteroid belt in early 2016, this object had a strange orbit and a tail of dust that resembled a comet. Through a careful analysis of telescopic imagery, scientists identified multiple showers of debris shooting up from its surface, the sort that could have only been produced by an impact.
What they had stumbled across was not a comet, but the immediate aftermath of an asteroid’s assassination.
"Knowing what types of impacts cause deflections and disruption is key to Earth’s protection from errant asteroids. That makes the demise of P/2016 G1 a vital source of information," said Megan Bruck Syal, a planetary defense researcher at the Lawrence Livermore National Laboratory.


New research by LLNL shows that Jupiter’s stripes, caused by the planet’s zonal winds, only descend to 3,000 kilometers in the atmosphere. Image courtesy of NASA
Jupiter’s stripes are only skin deep
NASA launched the Juno spacecraft toward Jupiter in 2011, and after a lengthy four-year trek, it finally began its orbit of the gaseous planet.
The Juno mission’s overarching goal is to understand how Jupiter formed, which could tell us about the enigmatic origins of our solar system. Juno is transmitting a boatload of data back to Earth. With that data, scientists aim to unlock the secrets of this massive, swirling ball of gas, including its bands of color.
Until Juno, scientists weren’t sure what lies below its impressionist painting-like surface. In 2016, using radio signals that mapped the distribution of mass in Jupiter’s atmosphere, Juno found that below a depth of 3,000 kilometers (4 percent of Jupiter’s diameter), the winds just stop.
What makes Jupiter’s winds different from Earth’s is that they’re not actually a gas, they’re an exotic fourth state of matter called plasma. Plasmas are ionized gases, meaning they conduct electricity and are strongly affected by magnetic fields. Deeper into the planet, where the pressure is higher, the atmosphere becomes more conducting and is more strongly influenced by the planetary magnetic field. It’s also the same place where the stripes stop.
“Most of the universe is in a plasma,” said Jeffrey Parker, a scientist at Lawrence Livermore, “so to understand the universe, it makes sense to start with the plasma."


LLNL researchers used the Omega Laser Facility at the University of Rochester’s Laboratory for Laser Energetics to make highly detailed measurements of laser-heated plasmas. Courtesy of J. Adam Fenster/University of Rochester
Energize me
New research from the University of Rochester and Lawrence Livermore (LLNL) could lead to more accurate computer models for simulations of laser-driven implosions. Researchers at the university’s Laboratory for Laser Energetics (LLE), along with their colleagues at LLNL and the Centre National de la Recherche Scientifique, have demonstrated how laser beams modify the conditions of the underlying plasma as they move through it, affecting the transfer of energy in fusion experiments.
In laser-driven inertial confinement fusion (ICF) experiments, short beams consisting of intense light pulses are used to heat and compress hydrogen fuel cells. Ideally, this process results in a greater release of energy than the amount of energy used to heat the cells.
Laser-driven ICF experiments require that many laser beams propagate through a plasma of free-moving electrons and ions to deposit their radiation energy precisely at their intended target. As the beams move through the plasma, they interact with it in ways that can complicate the intended result.
To accurately model the laser-plasma interaction, scientists need to know exactly how the energy from the laser beam interacts with the plasma and that’s what the team achieved.

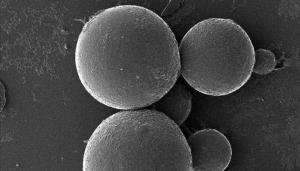
Helium ion microscopy images of microbe capsules at different magnification. At high magnification, encapsulated microbes are visible on the surface. The average bead size is 59 micrometers.
Waste not, want not
To increase the U.S. supply of rare earth elements, a Critical Materials Institute team led by Lawrence Livermore is using microbe beads to recover rare earth elements from consumer electronic waste such as cellphones.
Rare earth elements (REEs) such as neodymium and dysprosium are essential for technologies like solar and wind energy, advanced vehicles and modern electronics like smartphones. They comprise 17 elements in the periodic table. Despite their name, the rare earths (except for promethium) are not all that rare and found in relatively high concentrations across the globe. However, because of their geochemical properties, they are seldom discovered in easily exploitable deposits. Once mined, it usually takes extensive (and expensive) chemical processing to isolate the individual REEs. As a result, the cost of REEs is climbing while they become more essential for American competitiveness in the clean energy industry, as well as in many devices important to a high-tech economy and national security.
The new method could be used to recover REE’s from end of life products that use neodymium (NdFeB) magnets. These permanent rare-earth magnets are commonly used in wind turbines, hybrid and electric vehicles, household electrical appliances, computer hard disk drives and small consumer electronic devices, such as cellphones. The magnet has different life cycles depending on the application, from as short as 2-3 years for cellphones to 20-30 years in wind turbines.
“Recycling off REEs in this type of magnet from the end of life products will play an important and complementary role in the total supply of REEs in the future,” said Yongqin Jiao, LLNL bioscientist and co-lead author of the new study.


Images of the fungus Fusarium, a type of common plant pathogen that is a host for mitoviruses such as Narnaviridae, which are RNA viruses, and according to recent research, are highly abundant in soil. They have significant impacts on fungal fitness. Left photo by the USDA’s Agricultural Research Service. Right photo by the CDC
Going viral
Viruses impact nearly all organisms on Earth, with waves of influence in agriculture, health and biogeochemical processes.
However, very little is known about RNA viruses in the environment, and even less is known about their diversity and ecology in soil, one of the most complex microbial systems. But Lawrence Livermore National Laboratory (LLNL) scientists have helped discover that RNA viruses, specifically, are abundant and diverse in soil, where they prey upon organisms such as insects, nematodes and fungi.
“RNA viruses are known to target eukaryotes and some bacteria, but we know very little about them, their ecology and what their roles might be in soil,” said LLNL scientist Jennifer Pett-Ridge, a co-author on a paper appearing in the Proceedings of the National Academy of Sciences. “They are one of the great unknowns in the biology world and could be important predators in soil.
“They are great mobilizers of genetic information. We found that they are dynamic and different in different habitats in the soil. This means the dynamics of biotic relationships in soil are even more complicated than we’d known.”





