Implantable electrode coating good as gold
 (Download Image)
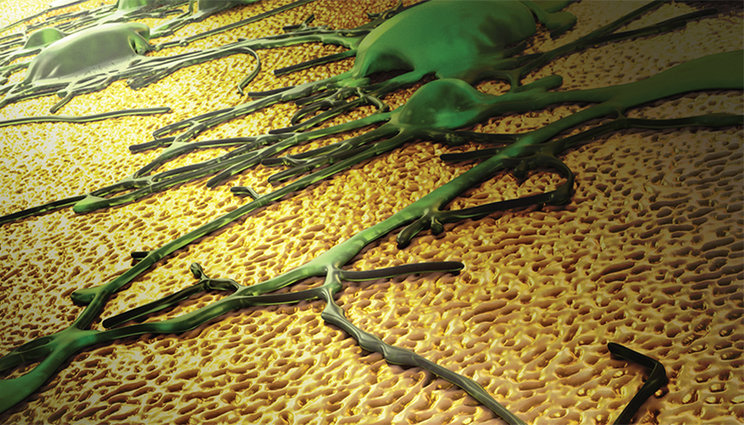
The image depicts a neuronal network growing on a novel nanotextured gold electrode coating. The topographical cues presented by the coating preferentially favor spreading of neurons as opposed to scar tissue. This feature has the potential to enhance the performance of neural interfaces. Image by Ryan Chen/LLNL.
(Download Image)
The image depicts a neuronal network growing on a novel nanotextured gold electrode coating. The topographical cues presented by the coating preferentially favor spreading of neurons as opposed to scar tissue. This feature has the potential to enhance the performance of neural interfaces. Image by Ryan Chen/LLNL.
A team of researchers from Lawrence Livermore and UC Davis have found that covering an implantable neural electrode with nanoporous gold could eliminate the risk of scar tissue forming over the electrode’s surface.
The team demonstrated that the nanostructure of nanoporous gold achieves close physical coupling of neurons by maintaining a high neuron-to-astrocyte surface coverage ratio. Close physical coupling between neurons and the electrode plays a crucial role in recording fidelity of neural electrical activity. The findings are featured on the cover of the journal Applied Materials & Interfaces.
Neural interfaces (e.g., implantable electrodes or multiple-electrode arrays) have emerged as transformative tools to monitor and modify neural electrophysiology, both for fundamental studies of the nervous system, and to diagnose and treat neurological disorders. These interfaces require low electrical impedance to reduce background noise and close electrode-neuron coupling for enhanced recording fidelity.
Designing neural interfaces that maintain close physical coupling of neurons to an electrode surface remains a major challenge for both implantable and in vitro neural recording electrode arrays. An important obstacle in maintaining robust neuron-electrode coupling is the encapsulation of the electrode by scar tissue.
Typically, low-impedance nanostructured electrode coatings rely on chemical cues from pharmaceuticals or surface-immobilized peptides to suppress glial scar tissue formation over the electrode surface, which is an obstacle to reliable neuron−electrode coupling.
However, the team found that nanoporous gold, produced by an alloy corrosion process, is a promising candidate to reduce scar tissue formation on the electrode surface solely through topography by taking advantage of its tunable length scale.
"Our results show that nanoporous gold topography, not surface chemistry, reduces astrocyte surface coverage," said Monika Biener, one of the LLNL authors of the paper.
Nanoporous gold has attracted significant interest for its use in electrochemical sensors, catalytic platforms, fundamental structure−property studies at the nanoscale and tunable drug release. It also features high effective surface area, tunable pore size, well-defined conjugate chemistry, high electrical conductivity and compatibility with traditional fabrication techniques.
"We found that nanoporous gold reduces scar coverage but also maintains high neuronal coverage in an in vitro neuron-glia co-culture model," said Juergen Biener, the other LLNL author of the paper. "More broadly, the study demonstrates a novel surface for supporting neuronal cultures without the use of culture medium supplements to reduce scar overgrowth."
UC Davis researchers include Christopher Chapman, Hao Chen, Marianna Stamou, Pamela Lein and Erkin Seker.
The LLNL portion of the work was funded by the UC Lab Fees Research Program.
Contact
 Anne M. Stark
Anne M. Stark
[email protected]
(925) 422-9799
Related Links
Nanporous Gold as a Neural Interface Coating: Effects of Topography, Suface Chemistry, and Feature Size"Nanoscale Synthesis and Characterization Laboratory
It’s nanotubular: New material could be used for energy storage and conversion
Tags
Bioscience and BioengineeringNeural devices
Engineering
Physical and Life Sciences
Featured Articles







