Research reveals how humidity affects atmospheric corrosion of aluminum metal
 (Download Image)
(Download Image)
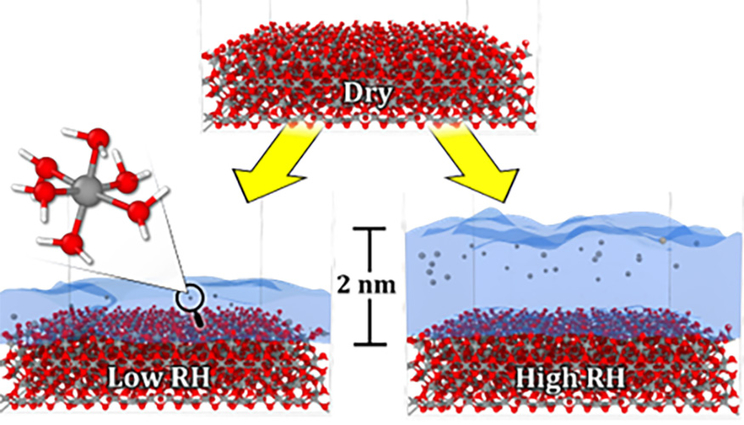
Metal oxides exposed to air form a nanoscale water film over the surface whose thickness depends on relative humidity. All-atom molecular dynamics simulations predict that dissolved aluminum ions inside the film will localize near the water’s surface, where they diffuse much more quickly than they do closer to the oxide. Lowering humidity increases confinement with the thin film, slowing the rate of atmospheric corrosion.
Scientists at Lawrence Livermore National Laboratory (LLNL) performed simulations using the Lab's supercomputer Ruby to uncover physical mechanisms that explain why humidity controls the rate of atmospheric corrosion of aluminum metal. Their research is featured in the ACS Journal of Applied Materials and Interfaces.
Accurate predictions of aluminum component lifetimes depend on assessments of corrosion rates. Engineering-scale models used for making system-level lifetime predictions are expressed in terms of coupled physical and chemical mechanisms including sorption, transport and chemical reactions. These mechanisms are inherently multiscale, which complicates both model form development and calibration.
Rates for atmospheric aluminum corrosion depends on relative humidity, which measures the amount of water present as vapor around the part. Understanding which processes give rise to these rate effects can help constrain the form of engineering lifetime models in terms of fundamental physical parameters.
When bare aluminum surfaces are exposed to air, they quickly react to form aluminum oxide. Water vapor in the surrounding humid air then adsorbs onto these oxide surfaces forming a nanoscopic film whose thickness depends on the relative humidity. Condensed surface water provides a medium for metal ions to dissolve and move through diffusion, which is important in the formation and growth of corrosion pits, but the confined nanoscopic dimensions can induce unusual effects.
To better understand how aluminum ions behave under confinement in water on surfaces, the team turned to all-atom molecular dynamics (MD) simulations for insights. MD makes comparatively few assumptions regarding how atoms interact and directly simulates a trajectory of atomic motions that can be post-processed to obtain material property data.
“Because diffusive transport is a comparatively slow process, we had to carefully select how to model atomic interactions,” said LLNL scientist Matt Kroonblawd, a co-author of the study. “Classical reactive molecular dynamics offers a desirable trade-off between accuracy and accessible time scales. Using reactive MD meant that we did not need to assume the structure of the aqueous aluminum species or the specific chemistry of the oxide surface.”
From their simulations, the team observed that aluminum ions tend to localize near the air-water interface and were completely absent near the oxide. This phenomenon was attributed to both the surface polarization of the water film and the rigid ice-like phase of water that forms near the oxide surface.
Interplay between these two interfacial phenomena resulted in height-dependent transport properties within the water film. Atoms diffuse very slowly near the oxide interface and have increasing diffusivity as the air-water interface is approached. Thickness of the surface water depends on relative humidity, which correlates these nanoscale confinement effects with empirically measured rates for atmospheric corrosion of aluminum.
“Confinement effects within oxide-adsorbed water films are well-documented in the literature, but this new insight on their direct impact on aqueous ion transport is tremendously useful in understanding the mechanisms of atmospheric corrosion,” explained LLNL scientist Jeremy Scher, lead author of the study.
The consequences of nanoscale confinement on corrosion rates became clearly apparent when the team upscaled their MD results to the continuum scale. A reductionist 1-dimensional continuum model of an aluminum corrosion pit was developed, which incorporated the ion diffusion coefficients calculated from the MD simulations. This simple model showed that corrosion rates can be diffusion limited under atmospheric conditions and are therefore strongly influenced by relative humidity.
“The results from this study highlight how essential it is to capture unusual nanoscale effects and their dependence on humidity when modeling atmospheric corrosion at larger length scales,” Scher said.
This study is part of a new multiscale modeling effort to extend the Reaction-Sorption Transport and Mechanics (or ReSorT-M) toolkit, a modeling framework used by the Materials Aging and Compatibility (MAC) group to help inform engineering decisions in Weapons and Complex Integration programs.
“Accurately determining the water layer’s thickness on the metal surface and understanding how humidity and temperature affect its properties are crucial for assessing corrosion concerns for programmatic needs,” said LLNL scientist Sylvie Aubry, a co-author in the study, and the ReSorT-M team lead.
LLNL researchers Stephen Weitzner, Tae Wook Heo, Yue Hao, Stephen Castonguay and Susan Carroll also contributed to this work.
Contact
 Anne M. Stark
Anne M. Stark
[email protected]
(925) 422-9799
Related Links
ACS Journal of Applied Materials and Interfaces.Tags
Advanced Materials and ManufacturingMaterials Science
Physical and Life Sciences
Featured Articles







