Breaking down the barriers in all solid-state batteries
 (Download Image)
(Download Image)
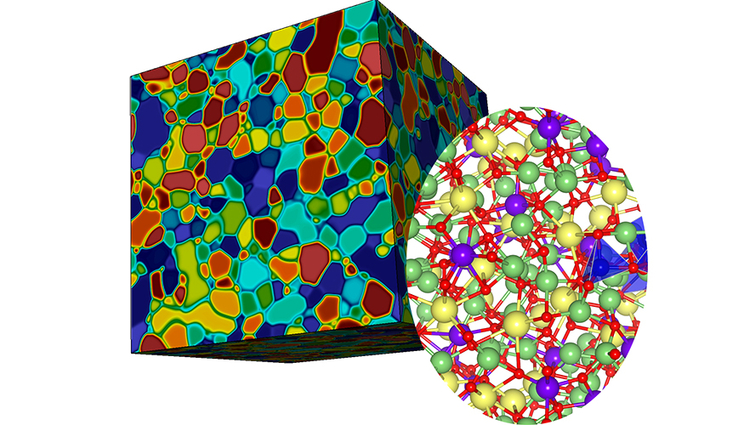
The multiscale model incorporates both microstructural (left) and atomistic (right) simulations to understand barriers to ion transport in solid-state battery materials. Image by Brandon Wood, Tae Wook Heo and Sabrina Wan/LLNL.
Solid electrolytes may overcome key technological hurdles associated with the narrow electrochemical and thermal stability of conventional lithium (Li)-ion and sodium (Na)-ion batteries.
However, many solid electrolytes — ceramics in particular — also suffer from poor cycling issues and limitations in their ability to efficiently transport ions. These limitations often stem from interfaces and other features that make up the microstructure of the material, which in turn depends on how it is processed.
Lawrence Livermore National Laboratory (LLNL) scientists in collaboration with San Francisco State University and the The Pennsylvania State University have developed a broad suite of multiscale simulation capabilities to help identify, assess and overcome microstructural impacts on ion transport in solid electrolytes. The research appears in the journal npj Computational Materials.
“We’ve come up with a powerful new computational modeling capability that can offer fundamental scientific understanding and practical design guidance not only to the energy storage research community but also to the materials processing community,” said Tae Wook Heo, LLNL scientist and lead author of the paper.
Microstructural features that appear unavoidably in practical solid-state materials — including defects, structural disorder and networks of internal interfaces — have a significant impact on the actual transport properties and battery lifetime. These features also introduce inhomogeneity in mechanical properties, which can have additional impacts on cyclability.
In the new research, the team looked to attain a better understanding of the detailed relationship between microstructure and ionic transport properties. According to Heo, this knowledge is critical to develop synthesis and processing pathways for viable solid electrolyte materials that retain high ionic conductivity.
“The current interest in solid-state batteries and processing science makes this work especially timely and impactful,” said LLNL project lead and co-author Brandon Wood.
The newly developed multiscale modeling framework is able to access unprecedented complexity by connecting atomistic simulations of atomic disorder and heterogeneity to a microstructure model that incorporates grain boundaries and other interfaces. The resulting tool can explore the effects of interfaces on transport through both scales, replacing conventional approaches like simple circuit models that lack structural detail. Heo said the new tool provides insight for resolving long-standing debates about the importance of microstructure in ceramic solid electrolytes. The researchers were able to quantify the effects of grain boundaries on ionic transport and identified possible correlations to common modes of battery degradation.
Other LLNL co-authors include Marissa Wood, Tim Hsu, Sneha Akhade and Liwen (Sabrina) Wan.
The research is funded by the Department of Energy Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office through the Battery Materials Research program and LLNL’s Laboratory Directed Research and Development program.
Contact
 Anne M. Stark
Anne M. Stark
[email protected]
(925) 422-9799
Related Links
npj Computational MaterialsVehicle Technologies Office
Tags
Advanced Materials and ManufacturingMaterials Science
Physical and Life Sciences
Global Security
Featured Articles







