LAB REPORT
Science and Technology Making Headlines
Sept. 1, 2017


An artist’s depiction of the promise of carbon nanotube porins for desalination. The image depicts a stylized carbon nanotube pipe that delivers clean desalinated water from the ocean to a kitchen tap. Image by Ryan Chen/LLNL
Better than Mother Nature
For the first time, a bio-inspired carbon nanotube (CNT) water filter has been made that can transport water faster than its biological counterparts.
The work could lead to new desalination membranes and ways to purify water to meet ever increasing global demand for fresh water. Cells possess water selective proteins called aquaporins that rapidly transport water through a membrane while rejecting salt ions. Their efficiency is attributed to their narrow 0.3 nanometer channels, which force water molecules to travel in single file. Wider CNTs have been shown to enhance water transport before due to their smooth interiors, but none has been able to coerce water into single file to become a serious rival to aquaporins.
Aleksandr Noy, at Lawrence Livermore National Laboratory, and colleagues have discovered that short lengths of narrow CNTs -- dubbed CNT porins (CNTPs) -- can confine water into single-file chains and transport it faster than aquaporins. Moreover, these CNTPs also have a high degree of ion selectivity that can reject salt in water saltier than seawater.

In this figure, the upper layer shows the release location probability contours and the lower layer shows the geographic region surrounding the Diablo Canyon Power Plant. The actual release location is denoted by the red X.
Pinpointing the source
Using data collected during an atmospheric tracer experiment three decades ago at the Diablo Canyon Nuclear Power Plant on the Central Coast of California, tens of thousands of computer simulations and a statistical model, researchers at Lawrence Livermore have created methods that can estimate the source of an atmospheric release with greater accuracy than before.
The methods incorporate two computer models: the U.S. Weather Research and Forecasting (WRF) model, which produces simulations of wind fields, and the community FLEXPART dispersion model, which predicts concentration plumes based on the time, amount and location of a release. Using these models, atmospheric scientists Don Lucas and Matthew Simpson at LLNL's National Atmospheric Release Advisory Center (NARAC) have run simulations developed for national security and emergency response purposes.
In 1986, to assess the impact of a possible radioactive release at the Diablo Canyon plant, Pacific Gas & Electric (PG&E) released a non-reactive gas, sulfur hexafluoride, into the atmosphere and retrieved data from 150 instruments placed at the power plant and its surrounding area. This data was made available to LLNL scientists, presenting them with a rare and valuable opportunity to test their computational models by comparing them with real data.
"Occasionally, the models we use in NARAC undergo development and we need to test them," Lucas said. "The Diablo Canyon case is a benchmark we can use to keep our modeling tools sharp.”

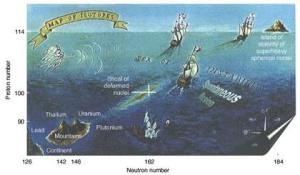
The island of stability is a region on the periodic table where superheavy elements would be more stable and last for more than a few milliseconds.
It’s elemental
Atom smashers have recently produced some new elements. Their appearance was brief, but long enough to fill out the periodic table.
Lawrence Livermore’s Mark Stoyer explains to Big Picture Science how new elements form and how, often, they only last for a millisecond. He discusses how the four newest elements on the periodic table were named.
And he discusses the elusive island of stability, which is a term in nuclear physics that refers to the possible existence of a region beyond the current periodic table where new superheavy elements with special numbers of neutrons and protons would exhibit increased stability. Such an island would extend the periodic table to even heavier elements and support longer isotopic lifetimes to enable chemistry experiments.
Listen to Stoyer on Big Picture Science, the science radio show & podcast of the SETI Institute.


A cutaway shows the interior of Neptune (left). Image by Greg Stewart/SLAC National Accelerator Laboratory
Diamonds are forever in the center
Scientists have just found out that it rains solid diamonds on Uranus and Neptune, thanks to the extreme pressures on those planets.
The gems form due to the oceans filled with hydrocarbon that rap around the solid cores of these giant planets, and scientists have long thought that extreme pressures might split the molecules into hydrogen and carbon.
As a result, the separated carbon crystallizes into diamonds, which then sink like rain through the “ocean” until they land on the solid core beneath. Up until now, however, scientists were unable to prove that this was the case. In a study published in the journal Nature Astrophysics, Lawrence Livermore researchers and collaborators were able to recreate this “diamond ring” with lasers and plastic.
Scientists had tried to replicate such conditions before, but imitating the insane pressure on these planets was difficult, as Neptune and Uranus have many times the mass of Earth. So researchers used two types of lasers to produce shock waves, which were then driven through polystrene plastic made of hydrogen and carbon.


A new 3D printing technique, developed at Lawrence Livermore, could allow scientists to print glass that incorporates different refractive indices in a single flat optic, making finishing cheaper and easier. Photos by Jason Laurea/LLNL
Through the looking glass
Lawrence Livermore scientists and academic collaborators have demonstrated the synthesis of transparent glass through 3D printing, a development that could lead to altering the design and structure of lasers and other devices that incorporate optics.
A team of LLNL researchers, along with scientists from the University of Minnesota and Oklahoma State University, report the creation of 3D-printed transparent glass components in the latest issue of Advanced Materials. In the paper, the researchers describe a 3D printing technique enabling glass structures and composition gradients previously impossible through conventional manufacturing processes.
"The Lab always looks for different ways to create new materials for optical applications," said LLNL chemical engineer and project lead Rebecca Dylla-Spears. "We're not going to replace the optical materials made through traditional means, but we're trying to impart new functionality using additive manufacturing. This is the first step to being able to print compositionally graded glass optics."





