Lawrence Livermore scientists discover bacterial resistance to improve biofuel production
 (Download Image)
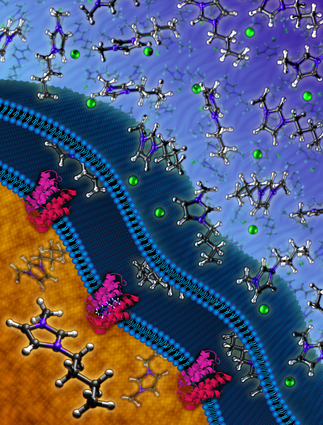
Ionic liquids (molten salts) are important solvents in the microbial production of biofuels, but can inhibit microbial growth. Lawrence Livermore researchers discovered a resistance mechanism in a rainforest soil bacterium that enables E. coli to grow and produce biofuel in the presence of ionic liquids at levels that otherwise would be toxic to native strains.
(Download Image)
Ionic liquids (molten salts) are important solvents in the microbial production of biofuels, but can inhibit microbial growth. Lawrence Livermore researchers discovered a resistance mechanism in a rainforest soil bacterium that enables E. coli to grow and produce biofuel in the presence of ionic liquids at levels that otherwise would be toxic to native strains.
New research by scientists from Lawrence Livermore National Laboratory in conjunction with the Joint BioEnergy Institute (JBEI) suggests that a type of bacterial resistance may provide more efficient production of biofuels.
The team identified the genetic origin of bacterial resistance to an ionic liquid (a salt in the liquid state), which they successfully introduced into a strain of E. coli bacteria for the production of advanced biofuels. The ionic liquid resistance is based on a pair of genes discovered in a microbial species native to a tropical rainforest in Puerto Rico.
"Ionic liquids are used as potent solvents to extract cellulose from biomass, so that it can be broken down to sugars used by microbes to make advanced biofuels -- new liquid fuels that go beyond ethanol and can replace gasoline or diesel," said Michael Thelen, an LLNL biochemist who also is part of JBEI's Deconstruction Division.
The team identified two genes in Enterobacter lignolyticus, a rainforest soil bacterium that is tolerant to specific ionic liquids, and transferred them as part of a genetic module into an E. coli biofuel host. The genetic module conferred the tolerance needed for the E. coli to grow well in the presence of toxic concentrations of ionic liquids. As a result, production of a terpene-based biofuel was enhanced.
The research appears in the March 26 edition of the journal, Nature Communications.
At JBEI, researchers have previously engineered strains of E. coli bacteria to digest the cellulosic biomass of switchgrass, a perennial grass that thrives on land not suitable for food crops, and convert its sugars into biofuels and chemicals. However, the ionic liquids used to make the switchgrass digestible proved to be too toxic for the E. coli and had to be completely removed through several washings prior to fermentation.
"The new genes confer to E. coli the ability to grow in the presence of normally toxic levels of an ionic liquid, making it possible to produce biofuels more efficiently," Thelen said. "The consequences of this study pave the way for further improvements in the microbial conversion of biomass to biofuels."
In this latest study, JBEI researchers used an approach devised by lead author and Basel University graduate student and LLNL guest researcher Thomas Ruegg to rapidly pinpoint the genes responsible for ionic liquid resistance in the genomic DNA of Enterobacter lignolyticus. "This genetic module encodes both a membrane transporter and its transcriptional regulator," Ruegg said. "While a pump exports ionic liquids, the substrate-inducible regulator maintains the appropriate level of this pump so that the microbe can grow normally either in the presence or absence of ionic liquid."
The results are likely to eliminate a bottleneck in JBEI's biofuels production strategy, which relies on ionic liquid pretreatment of cellulosic biomass. The research also demonstrates how the adverse effects of ionic liquids can be turned into an advantage, by inhibiting the growth of other bacteria. This research was funded by the DOE Office of Science.
Contact
 Anne M. Stark
Anne M. Stark
[email protected]
(925) 422-9799
Related Links
Joint BioEnergy InstituteMichael Thelen
Lawrence Livermore work may improve the efficiency of the biofuel production cycle
"From Data to Discovery"




