Lab team receives grant to study marine microbes' link to carbon cycle
 (Download Image)
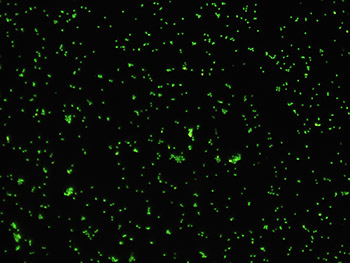
A marine microbe with DNA that has been stained with the fluorescent dye "SYBRgreen."
(Download Image)
A marine microbe with DNA that has been stained with the fluorescent dye "SYBRgreen."
And now he will have the opportunity after he and LLNL colleague Jennifer Pett-Ridge and collaborators from Oak Ridge National Laboratory and Oregon State University received a nearly $1 million grant from the Gordon and Betty Moore Foundation Marine Microbiology Initiative.
High precision isotope techniques and molecular biology methods will be combined in new ways to characterize the flow of nutrients in marine microbial ecosystems to identify which microbes prefer which types of food molecules and how microbial food web interactions influence the flow and fate of ocean carbon.
Nearly 50 gigatons of carbon, or about 45 percent of the Earth's primary production, are fixed within the ocean annually. Marine microbial communities are responsible for the processing of nearly half of this carbon. The relationship between environmental factors and the microbial control of carbon sequestration, respiration, or export to higher trophic levels of marine food webs is not well understood, given the complexity and high variability of microbial communities across temporal, physical and chemical gradients in the ocean.
The initial experiments will focus on seawater collected from Monterey Bay. Monterey Bay Aquarium Research Institute (MBARI) scientists have compiled extensive long-term datasets detailing the geochemical, physical and biological nature of seawater collected from multiple sampling sites within this ecosystem (e.g., temperature, salinity, transmissibility, chlorophyll levels, nitrate concentrations, bacterial community composition, algal bloom dynamics).
"These data will be critical in directing our experimental design and interpreting the results of different treatment tests," Mayali said. "We expect that the technical approach we develop to describe the Monterey Bay system will be adaptable for estuarine and offshore habitats as well."
The work will use LLNL's NanoSIMS, one of only five in the world used for biological applications, and Oak Ridge's Thermo LTQ-Orbitrap Velos (the most precise mass spectrometer for proteomics). Both instruments and associated infrastructures are operational for microbial systems.
"By combining traditional stable isotopic probing (SIP) approaches with advanced mass-spectrometry (MS) techniques, our team has developed novel, high-throughput methods for the accurate and sensitive measurement of species-specific functions within natural microbial communities," he said.
The team's goals are:
- Characterize taxa-specific preferences for the most common carbon resources within a well characterized coastal upwelling ecosystem (e.g., proteins vs. lipids vs. polysaccharides).
- Identify functional guilds of microbes responsible for the processing of organic carbon.
- Understand how abiotic factors (nitrogen and phosphorus inputs, irradiation, temperature) influence the utilization of organic matter.
- Define species-specific interactions between phytoplankton primary producers and bacterial consumers and the substrates mediating those interactions.
- Develop a streamlined approach to combining isotope labeling experiments with 16S rRNA and proteomic analysis that can be used broadly in a range of marine habitats.




