LAB REPORT
Science and Technology Making Headlines
Feb. 7, 2020

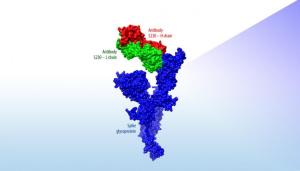
This figure shows how S230, an antibody known to have neutralizing activity against the Severe Acute Respiratory Syndrome coronavirus, is predicted to interact with an initial model of a surface protein on the novel coronavirus. This computational estimate can quickly provide researchers with structural insights without waiting for time-consuming X-ray crystallography images of the actual protein.
Modeling a virus
As global concern continues to rise about a novel coronavirus spreading from China, a team of Lawrence Livermore National Laboratory researchers has developed a preliminary set of predictive 3D protein structures of the virus to aid research efforts to combat the disease.
The team’s predicted 3D models, developed over the past week using a previously peer-reviewed modeling process, are based on the genomic sequence of the novel coronavirus and the known structure of a protein found in the virus that causes Severe Acute Respiratory Syndrome (SARS), also a coronavirus that closely resembles the new virus.
“A major part of the value of these new structural models is that they present the predicted protein in complex with SARS-neutralizing antibodies,” said Adam Zemla, an LLNL structural biologist and mathematician. “This can be thought of as the first step for the global research community to identify and model how therapeutic antibodies can be designed to fight the novel coronavirus.”
Lab researchers are designing a diversity of protein models because the new coronavirus protein structure is not yet known, according to Daniel Faissol, a data scientist in the Lab’s Computational Engineering Division.


Climate change can cause glacier melt, as depicted in these images of Muir Glacier, Alaska in Aug. 13, 1941 and Aug. 13, 2004. Image courtesy of NASA.
Running red hot
There are dozens of climate models, and for decades they’ve agreed on what it would take to heat the planet by about 3 degrees Celsius. It’s an outcome that would be disastrous — flooded cities, agricultural failures, deadly heat — but there’s been a grim steadiness in the consensus among these complicated climate simulations.
Then last year, some of the models started running very hot. The scientists who hone these systems used the same assumptions about greenhouse-gas emissions as before and came back with far worse outcomes. Some produced projections in excess of 5 degrees C, a nightmare scenario.
“The question is whether they’ve overshot,” said Mark Zelinka, staff scientist at Lawrence Livermore National Laboratory.
In the next year, climate-modeling groups will peruse each other’s results to figure out how seemingly good improvements in cloud and aerosol science may have pushed the models into hotter states. These conversations happen in the open, through peer-reviewed journals, conferences and blog posts. The authors of the main U.N. climate-science reports will follow along and try to stitch together a big picture, for release in 2021.


Lead researcher Dave Soscia said the “brain-on-a-chip” device, designed and fabricated at Lawrence Livermore National Laboratory, was designed to be easily reproducible. The Lab has applied for a patent on the device and is looking to engage with potential collaborators to further develop it. Photos by Julie Russell/LLNL.
This is your brain, on a chip
Lawrence Livermore National Laboratory engineers and biologists have developed a “brain-on-a-chip” device capable of recording the neural activity of living brain cell cultures in three-dimensions, a significant advancement in the realistic modeling of the human brain outside of the body.
LLNL researchers report on the creation of a 3D microelectrode array (3DMEA) platform in which they were able to keep hundreds of thousands of human-derived neurons alive, networked and communicating in a 3D gel, while non-invasively recording their electrical spikes and bursts for up to 45 days using LLNL-developed, thin-film microelectrode arrays.
Researchers said the knowledge gained from the device could inform science in developing countermeasures for warfighters exposed to chemical or biological agents, model disease or infection, evaluate environmental toxins or aid in drug discovery, without the need for animal models.


A fluid that stiffens in response to a magnetic field is injected into the hollow struts and beams of a 3D-printed lattice. The material can be made stiff or flexible. Credit: Julie Mancini/LLNL
3D goes big, fast and strong
Three-dimensional printing has come a long way in the past 20 years.
There is a slew of research advances in 3D printing that are broadening the prospects of a technology once viewed as useful mainly for making small, low-quality prototype parts. Not only is 3D printing becoming faster and producing larger products, but scientists are coming up with innovative ways to print and are creating stronger materials, sometimes mixing multiple materials in the same product.
New resin-printing techniques stil arel emerging. The approach has garnered substantial interest from industry, says Christopher Spadaccini, a materials and manufacturing engineer at Lawrence Livermore National Laboratory. Spadaccini was a member of the team that published the idea last January. He thinks the technology has tremendous commercial potential because it has modest hardware requirements. “In the end, really, what you need is a halfway-decent projector and a rotating stage,” he says.
Aviation firms such as Boeing, Rolls Royce and Pratt & Whitney are using 3D printing to make metal parts, mainly for jet engines. It can be cheaper than milling metal blocks, and the intricate components often weigh less than their conventionally made counterparts.
But 3D-printed metals are prone to defects that can weaken the final products. Spadaccini and others are trying to use arrays of sensors and high-speed cameras to watch for irregularities such as hotspots of heat or strain — and then make adjustments in real time, he says.

Three-dimensional-printed electrodes boost charge rate and capacity of electrical storage devices. Image by Adam Connell/LLNL
A boost for energy storage
A team of Lawrence Livermore National Laboratory (LLNL) scientists and collaborators have developed a new class of aerogel electrodes with a simultaneous boost in energy and power density. The research could be a boon for the energy storage industry.
“This is the first example in which we were able to boost both parameters in the same device,” said Swetha Chandraskaran, an LLNL material scientist and a co-author of a paper appearing in the journal Advanced Materials
Three-dimensional printing technologies have been extensively used in different research fields such as energy storage devices, catalysis, electronics, microfluidics and biotechnology. These printing technologies have enabled the creation of unique material and device structures that cannot be achieved by conventional methods.
By exploring new complex architectures, 3D‐printed materials can achieve novel and/or improved functionality. Direct ink writing (DIW) is one of the most commonly used 3D‐printing techniques. It offers great flexibility in the material (ink) selection and has been recently applied to prepare electrodes for electrochemical energy storage devices, including lithium ion batteries, sodium ion batteries, lithium sulfur batteries, lithium metal batteries and supercapacitors.





