LAB REPORT
Science and Technology Making Headlines
March 1, 2024


LLNL has developed a new solid-state electrolyte that is a safer alternative the current technology in lithium-ion batteries. Image by Adobe Stock.
A new take on lithium ion batteries
Using computational science, researchers at the Korea Institute of Science and Technology (KIST) and Lawrence Livermore National Laboratory have developed a fluorine-substituted high-voltage stable chloride-based solid-state electrolyte.
This new material is expected to be non-flammable and a safer alternative to liquid electrolytes commonly found in lithium-ion batteries.
To improve the high-voltage stability of chloride-based solid electrolyte, the research team proposed the optimal composition and design principle of chloride-based solid electrolyte substituted with fluorine, which has strong chemical bonding ability.
For the proposed strategy to achieve this goal, LLNL contributed by utilizing their supercomputing resources for calculations and subsequent experimental validations were conducted at KIST. The collaborative research team adopted a cost-effective and time-saving strategy, wherein computational science guides the initial material design, followed by rigorous laboratory validation.


An artist’s view of small-diameter carbon nanotubes that pass through water molecules (red and white) and reject ions (blue). High permselectivity of small-diameter nanotubes can enable advanced water desalination technologies. illustration concept: A. Noy, T. A. Pham, Y. Li, Z. Li, F. Aydin (LLNL). Illustration by Ella Maru Studios.
Small pores pack big punch
Vertically aligned carbon nanotube (VaCNT) membranes can be used to clean or desalinate water with a high flow rate and low pressure. Recently, researchers from the Karlsruhe Institute of Technology (KIT) and Lawrence Livermore conducted steroid hormone adsorption experiments to investigate the interaction of forces in microscopic pores.
They discovered that VaCNTs with certain pore geometry and surface structure are suitable for use as highly selective membranes.
In studies using steroid micropollutants, KIT researchers investigated why VaCNT membranes make excellent water filters. They utilized membranes made by Lawrence Livermore National Laboratory (LLNL). The finding: VaCNT's low adsorption, or surface deposition, is useful for highly selective membranes that target specific molecules.
At LLNL, Francesco Fornasiero and his colleagues created the membranes. The most recent analytical equipment at KIT was used to conduct and assess the micropollutant tests.

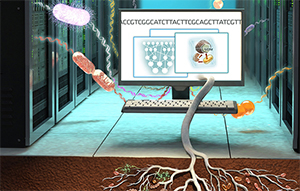
Microbe models leverage extensive genomic data to power soil carbon simulations. Illustration by Victor O. Leshyk.
Getting to the root of the matter
Climate models are essential to predicting and addressing climate change, but can fail to adequately represent soil microbes, a critical player in ecosystem soil carbon sequestration that affects the global carbon cycle.
A team of scientists led by Lawrence Berkeley National Laboratory (Berkeley Lab) and including Lawrence Livermore researchers has developed a new model that incorporates genetic information from microbes. This new model enables the scientists to better understand how certain soil microbes efficiently store carbon supplied by plant roots, and could inform agricultural strategies to preserve carbon in the soil in support of plant growth and climate change mitigation.
This new observation provides a basis for improving how root-microbe interactions are represented in models, and enhances the ability to predict how microbes impact changes to the global carbon cycle in climate models.


As described in the paper, the LLNL team, along with their collaborators from Harvard University, North Carolina State University, and the University of Pennsylvania, used a direct ink writing printing technique to build a variety of light-responsive objects, including cylinders that could roll, asymmetric “crawlers” that could go forward, and lattice structures that oscillated. By combining shape morphing with photoresponsivity, researchers said the new type of material could change the way people think about machines and materials. Photos courtesy: Michael Ford/LLNL.
Shapeshifting comes to light
Researchers at Lawrence Livermore National Laboratory have furthered a new type of soft material that can change shape in response to light, a discovery that could advance “soft machines” for a variety of fields, from robotics to medicine.
The novel material, called a liquid crystal elastomer (LCE), is made by incorporating liquid crystals into the molecular structure of a stretchable material. Adding gold nanorods to the LCE material, scientists and engineers created photo-responsive inks and 3D printed structures that could be made to bend, crawl, and move when exposed to a laser that causes localized heating in the material. The results were recently published online by the journal Matter.
As described in the paper, the LLNL team, along with their collaborators from Harvard University, North Carolina State University and the University of Pennsylvania, used a direct ink writing printing technique to build a variety of light-responsive objects, including cylinders that could roll, asymmetric “crawlers” that could go forward, and lattice structures that oscillated. By combining shape morphing with photoresponsivity, researchers said the new type of material could change the way people think about machines and materials.


Lawrence Livermore is participating in a project that would explore wind energy technologies off the New England coast. Photo courtesy of NOAA Fisheries, National Oceanic and Atmospheric Administration.
Bring on the wind
The Wind Forecast Improvement Project 3 (WFIP3) is venturing into offshore wind energy exploration off the New England coast. The project seeks to address the complexities of forecasting offshore winds.
The project is deploying instruments across six land-based sites, with Argonne instrumentation at Martha’s Vineyard, Cape Cod, Nantucket Island and Rhode Island. A barge, equipped with instrumentation, will soon be moored in the waters where offshore wind turbines are being installed.
The instrumentation will provide detailed ocean observations from the surface to the top of the boundary layer. These observations will be crucial in refining and validating forecast models, ultimately advancing the understanding of offshore wind energy resources.
The project is funded by the U.S. Department of Energy (DOE) Wind Energy Technology Office, with the federal team including Argonne National Laboratory, Pacific Northwest National Laboratory, the National Renewable Energy Laboratory, Lawrence Livermore National Laboratory as well as the National Oceanic and Atmospheric Administration. The non-federal partner team is led by the Woods Hole Oceanographic Institution and includes several university partners.





