LAB REPORT
Science and Technology Making Headlines
March 29, 2024


Supercomputer simulations predicting the synthesis pathways for the elusive BC8 “super-diamond,” involving shock compressions of diamond precursor, inspire ongoing Discovery Science experiments at NIF. Image by Mark Meamber/LLNL.
Diamonds are a lab’s best friend
Diamonds are the hardest natural substance on Earth, but that may not necessarily be true on carbon-rich exoplanets where certain temperature and pressure conditions create an ultra-dense form of carbon called eight-atom body-centered cubic, or BC8, also known as super-diamond.
Scientists have known about BC8 for decades but have never observed the atomic structure, but Lawrence Livermore scientists and collaborators have now used a supercomputer to create simulations that help understand how BC8 could form.
The major finding helps explain why previous attempts at finding BC8 were unsuccessful because it turns out that the super-diamond can only form in a very narrow band of pressure and temperature. The team is now busy exploring if these theoretical pathways can finally produce some very real-world results.


Cover cropping has a relatively high potential contribution to national soil-based CO2-removal efforts, due mainly to the large extent of land area that can be cover-cropped without interfering with cash-crop production. Cereal rye is by far the most widely planted cover crop currently in the United States.
Counting on Mother Nature
Fifty-five gigatons of greenhouse gases are released into the atmosphere each year, according to the United Nations. The vast majority of climate scientists agree that human activities have been the main driver of climate change, primarily due to the burning of fossil fuels, and that cutting emissions is necessary to curb warming trends.
The United States and North Carolina have goals to cut greenhouse gas emissions and reach “net-zero” by 2050. Completely eliminating greenhouse gas emitting activities would be an enormous challenge in our current economy. Instead, countries are hoping to fight climate change with a “net-zero” strategy: removing one molecule of greenhouse gas from the air for every molecule that's released, making the net emissions zero.
That can be achieved by utilizing cleaner technology and more efficient processes to reduce emissions, as well as capturing carbon from the atmosphere and storing it.
“One of the easiest ways to do it is using Mother Nature,” said Allegra Mayer, a researcher at Lawrence Livermore National Laboratory and co-author or the report Roads to Removal. “Plants just breathe carbon dioxide out of the air. It can be stored in soil, forests and cover crops.”
She says North Carolina forests are some of the best in the country for carbon-storage capability.

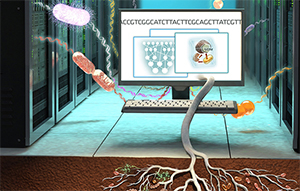
Microbe models leverage extensive genomic data to power soil carbon simulations. Illustration by Victor O. Leshyk.
Secrets beneath the surface
Climate models are essential to predicting and addressing climate change, but can fail to adequately represent soil microbes, a critical player in ecosystem soil carbon sequestration that affects the global carbon cycle.
A team of scientists from Lawrence Berkeley National Laboratory (Berkeley Lab) and Lawrence Livermore has developed a new model that incorporates genetic information from microbes. This new model enables the scientists to better understand how certain soil microbes efficiently store carbon supplied by plant roots, and could inform agricultural strategies to preserve carbon in the soil in support of plant growth and climate-change mitigation.
“Our research demonstrates the advantage of assembling the genetic information of microorganisms directly from soil. Previously, we only had information about a small number of microbes studied in the lab,” said Berkeley Lab postdoctoral researcher Gianna Marschmann. “Having genome information allows us to create better models capable of predicting how various plant types, crops, or even specific cultivars can collaborate with soil microbes to better capture carbon. Simultaneously, this collaboration can enhance soil health.”
The corresponding authors are Eoin Brodie of Berkeley Lab, and Jennifer Pett-Ridge of Lawrence Livermore, who leads the “Microbes Persist” Soil Microbiome Scientific Focus Area project that is funded by the DOE Office of Science in support of this work.
Soil microbes help plants access soil nutrients and resist drought, disease and pests. Their impacts on the carbon cycle are particularly important to represent in climate models because they affect the amount of carbon stored in soil or released into the atmosphere as carbon dioxide during the process of decomposition. By building their own bodies from that carbon, microbes can stabilize (or store) it in the soil, and influence how much, and for how long carbon remains stored belowground. The relevance of these functions to agriculture and climate are being observed like never before.


A photo taken by a scanning electron microscope shows a pit at the surface of an additively manufactured (3D-printed) stainless steel part. Image by Thomas Voisin/LLNL.
They are just a bunch of slags
Researchers at the Lawrence Livermore National Laboratory delved into the mysterious world of pitting corrosion in additively manufactured (3D-printed) stainless steel 316L in seawater and discovered that the key players in this corrosion drama are tiny particles called “slags.”
Stainless steel 316L is a popular choice for marine applications due to its combination of mechanical strength and corrosion resistance. This holds even more true after 3D printing, but even this resilient material isn’t immune to pitting corrosion.
Slags are produced by deoxidizers such as manganese and silicon. In traditional stainless steel 316L manufacturing, these elements are typically added before casting to bind with oxygen and form a solid phase in the molten liquid metal that can be easily removed post-manufacturing.
The LLNL researchers found these slags also form during laser powder bed fusion (LPBF) 3D printing but remain at the metal’s surface and initiate pitting corrosion.
“Pitting corrosion is extremely difficult to understand due to its stochastic nature, but we determined the material characteristics that cause or initiate this type of corrosion,” said LLNL researcher Shohini Sen-Britain. “While our slags looked different than what had been observed in conventionally manufactured materials, we hypothesized that they could be a cause of pitting corrosion in 316L.”


Simon Pang (left) and Buddhinie Jayathilake assemble and prepare a prototype bubble column electrobioreactor to test additively manufactured three-dimensional electrodes. Under their project, excess renewable electricity from wind and solar sources would be stored in chemical bonds as renewable natural gas. Photo by Nathan Ellebracht/LLNL.
A renewable natural gas breakthrough
Lawrence Livermore, Southern California Gas Company (SoCalGas) and Electrochaea are collaborating on an innovative research project that aims to develop a single-stage electro-bioreactor to transform excess renewable electricity and biogas into carbon-neutral synthetic biomethane, also known as renewable natural gas (RNG).
This approach could mark a significant advancement in power to gas technology and underscores the viability of potential for synthetic biomethane to help decarbonize natural gas infrastructure and its end uses from residential heating to manufacturing industries and transportation.
If developed at scale, this technology could increase the yield of RNG produced from carbon dioxide sources like anaerobic digesters, landfills, dairies, fermentation facilities or industrial processes.
“We believe this technology will help enable decarbonization of the natural gas grid infrastructure by providing a renewable source of natural gas,” said Simon Pang, a materials scientist in LLNL’s Materials Science Division, who heads the project. “This renewable natural gas can be moved and used in existing infrastructure, allowing the technology to be deployed soon to meet green energy demand. Moreover, by producing pipeline-quality renewable natural gas from biogas, we can increase the value of biogas and reduce the likelihood that it will be vented to the atmosphere, reducing greenhouse gas emissions and improving local air quality.”





