Going beyond Mother Nature’s molecules to target radioactive metals
 (Download Image)
(Download Image)
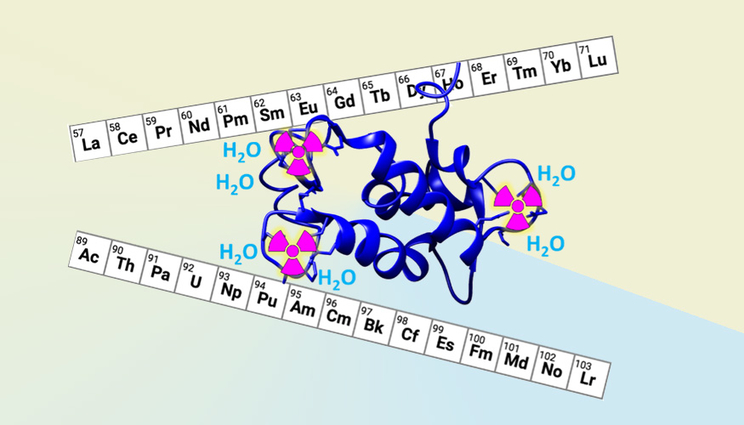
The natural protein lanmodulin (depicted in navy blue) was bioengineered by a collaboration of scientists from LLNL and Penn State University to better scavenge and separate elements found in nuclear waste, such as the actinides and their lanthanide fission products. Image by Gauthier Deblonde/LLNL.
Lawrence Livermore National Laboratory (LLNL) scientists and collaborators at Penn State University are improving natural molecules that would help target specific radioactive elements that are found in nuclear waste or used in nuclear medicine.
Even the most effective molecules found in nature, which underwent billions of years of evolution, can still be improved for non-natural applications. The team bioengineered nature’s most potent protein (lanmodulin) for lanthanides —natural elements used in numerous items like computer hard drives and magnets— to make it even more selective for actinide elements. Actinides are radioactive metals that are present in nuclear waste, such as uranium, plutonium and americium.
The research appears in the journal Chemical Science. The results improve the understanding of how natural compounds can interact with nuclear waste in the environment and could lead to new molecules for scavenging and detection of specific radioactive metals.
The team strategically designed, synthesized and characterized five variants of lanmodulin (LanM) to decipher and eventually improve its actinide-binding properties. Surprisingly, they found that the presence of water molecules that bridge the metal and protein molecule is particularly important for controlling the stability and metal preferences of the metal-protein complexes. This design principle allowed the scientists to improve the protein’s ability to discriminate between actinide and lanthanide elements.
Molecules that are selective for actinides over lanthanides are among the most coveted because these two families of elements are found in nuclear waste and separating them would allow for a more efficient management of radioactive materials. The team’s discovery could lead to drastically new separation systems for applications in nuclear waste and radiochemistry fields. LanM was discovered by the Penn State members of the team in 2018, and the LLNL-Penn State collaboration has been exploring applications of this peerless natural molecule in the field of nuclear sciences.
“This is the first study where someone made changes to lanmodulin to dissect and improve its metal binding properties,” said LLNL scientist Gauthier Deblonde, a co-lead author of the study. “As we were tuning the protein’s properties to target radioactive elements, we also learned a lot about the mechanisms by which it binds the metals.”
While classic molecules have a limited set of chemical interactions, the new research showed that macromolecules, such as proteins, have an extended repertoire of chemical interactions that scientists can fine-tune to target specific metals.
“This study uncovers yet another tool that this remarkable protein has at its disposal to discriminate between metals that differ from one another in only very subtle ways. This realization is an important step toward high-performance LanM-based separation methods and molecules custom-designed to bind specific medical isotopes,” said Joseph Cotruvo, Jr., Penn State assistant professor of chemistry and a co-lead author of the study.
Joseph Mattocks of Penn State also contributed to this work. The work is funded by the National Nuclear Security Administration’s Office of Defense Nuclear Nonproliferation Research and Development and the U.S. Department of Energy’s Basic Energy Sciences program.
Contact
 Anne M. Stark
Anne M. Stark
[email protected]
(925) 422-9799
Related Links
Chemical ScienceNational Nuclear Security Administration’s Office of Defense Nuclear Nonproliferation Research and Development
Tags
Physical and Life SciencesFeatured Articles







